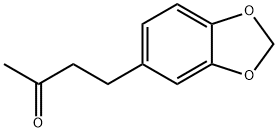
4-(3,4-Methylenedioxyphenyl)-2-butanone has an intensely sweet,
floral, slightly woody odor. May be prepared by condensation of
heliotropin with acetone, followed by hydrogenation in the presence of a palladium catalyst.
4-(3,4-Methylenedioxyphenyl)-2-butanone has an intensely sweet, floral, slightly woody odor reminiscent of raspberry,
cotton candy (i.e., candy floss) with a cassie, heliotrope association.
Has apparently not been reported to occur in nature.
By condensation of heliotropin with acetone, followed by hydrogenation in the presence of a palladium catalyst
ChEBI: 4-(3,4-Methylenedioxyphenyl)-2-butanone is a member of benzodioxoles.
Taste characteristics at 40 ppm: sweet, berry-like with spicy, jamy nuances
The oxygen-aromatic carbon link of aromatic ethers is generally biologically stable, and possible metabolites include the p-hydroxy derivative of the ether, the phenol or the p-hydroxyphenol (Williams, 1959). Ketones are not readily metabolized in the body. As a derivative of 2-butanone, piperonyl acetone might be expected to be partially reduced to the secondary alcohol and excreted as the glucuronide (Williams, 1959), since Saneyoshi (1911) isolated the glucuronide of 2-butanol from the urine of rabbits receiving methyl ethyl ketone.