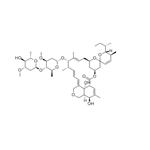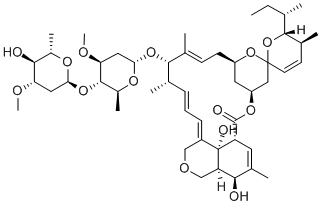
Abamectin
- Product NameAbamectin
- CAS71751-41-2
- MFC49H74O14
- MW887.11
- EINECS200-096-6
- MOL File71751-41-2.mol
Chemical Properties
| Melting point | 150-155°C |
| alpha | D +55.7 ±2° (c = 0.87 in CHCl3) |
| Boiling point | 717.52°C (rough estimate) |
| Density | 1.16 |
| vapor pressure | <2 x 10-7 Pa |
| refractive index | 1.6130 (estimate) |
| Flash point | 150 °C |
| storage temp. | Sealed in dry,Store in freezer, under -20°C |
| solubility | Soluble in DMSO |
| Water Solubility | 0.007-0.01 mg l-1 (20 °C) |
| Merck | 13,2 |
| InChIKey | GVWIWZFXCGTSLL-MSTMYQEVSA-N |
| CAS DataBase Reference | 71751-41-2(CAS DataBase Reference) |
| EPA Substance Registry System | Abamectin (71751-41-2) |
Avermectin is a kind of 16-membered ring macrolide compound which was first developed by the Kitasato University in Japan and Merck Company (United States). It has insecticidal, acaricidal, and nematicidal activity. It is produced by the fermentation of Streptomyces avermitilis. Natural avermectin containing eight component with four major components namely A1a, A2a, B1a and B2a with the total content being ≥80%; another four components corresponding to smaller proportion are A1b, A2b, B1b, and B2b with the total content of ≤20%. Currently commercialized avermectin pesticide has abamectin as the main insecticidal ingredient (Avermectin B1a + B1b with B1a being not less than 90% and B1b being less than 5%). It is calibrate by the content of B1a.
Safety Information
| Hazard Codes | T+,N |
| Risk Statements | 20-28-50 |
| Safety Statements | 36/37/39-45-60-61 |
| RIDADR | 2588 |
| WGK Germany | 3 |
| RTECS | CL1203000 |
| HazardClass | 6.1(a) |
| PackingGroup | II |
| Hazardous Substances Data | 71751-41-2(Hazardous Substances Data) |
| Toxicity | LD50 (technical grade) orally in sesame oil in mouse, rat: 13.5, 10.0 mg/kg; dermally in rabbit: >2000 mg/kg; LD50 in mallard duck, bobwhite quail: 84.6, >2000 mg/kg; LC50 (96 hr) in rainbow trout, bluegill: 3.6, 9.6 mg/l; LC50 (48 hr) in Daphnia magna: 0.34 mg/l (Merck Technical Data Sheet) |
MSDS
| Provider | Language |
|---|---|
| Affirm | English |
Usage And Synthesis
Abamectin is a kind of 16-membered ring macrolide compound which was first developed by the Kitasato University in Japan and Merck Company (United States). It has insecticidal, acaricidal, and nematicidal activity. It is produced by the fermentation of Streptomyces avermitilis. Natural Abamectin containing eight component with four major components namely A1a, A2a, B1a and B2a with the total content being ≥80%; another four components corresponding to smaller proportion are A1b, A2b, B1b, and B2b with the total content of ≤20%. Currently commercialized Abamectin pesticide has abamectin as the main insecticidal ingredient (Abamectin B1a + B1b with B1a being not less than 90% and B1b being less than 5%). It is calibrate by the content of B1a. Since 1991 when abamectin had entered into the market of pesticides in China, Abamectin pesticide has played an important role in the pest prevention and control system of China. Abamectin is currently produced by over tens companies in China with the currently marketed Abamectin series pesticides including abamectin, ivermectin and emamectin benzoate. In the late 1980s, Shanghai Institute of pesticides of China has isolated and screened the 7051 strain from Guangdong Jieyang soil. Identification analysis had proved that this strain is quite similar with S.avermitilis Ma-8460 and can produce a compound with the same chemical structure as Abamectin. In 1993, the new technology Development Corporation of Beijing Agricultural University had initiated research project for development and production of this drug. Abamectin is a new class of antibiotics characterized with a novel structure and dual application to both crops and livestock. With the improvement of people's living standards as well as the demanding for green good, bio-pesticides is quite preferred in current pesticide market. According to the pundits’ prediction, the 21st century will be the century of biological pesticides. It is reported that the European bio-pesticides sales increased from 100 million dollar (1997) to 160 million dollars in 2004. Abamectin is the most popular and highly competitive novel biological pesticide in currently bio-pesticide market.
Abamectin has its original drug be white or yellow-white crystalline powder with an active ingredient content being 75% to 80%, specific gravity being 1.16, melting point being 155~157 ℃, and vapor pressure being 2 × 10-7pa. At 21℃ It has a solubility being 7.8 g/liter in water, 100 mg/mL in acetone, 20 mg/mL in ethanol, 19.5 mg/mL in methanol, 10 mg/mL in chloroform, 6 mg/mL in ring hexane, 70 mg/mL in isopropyl alcohol, 0.5 mg/mL in kerosene, and 350 mg/mL in toluene. At room temperature is not easy to be decomposed. At 25 ℃, no decomposition is observed for its solution of pH6~9.
The Appearance of its preparation is light brown liquid. Its preparation can be stored stably at room temperature for more than 2 years.
Toxicity: According to Chinese pesticide toxicity grading standards, Abamectin belongs to highly toxic pesticides. For original drug; acute oral-rat LD50: l0 mg/kg; acute oral-mice LD50: 13 mg/kg; acute percutaneous administration-rabbits LD50: greater than 2000 mg/kg; acute percutaneous administration-rat LD50: greater than 380 mg/kg; acute inhalation-rat LC50 greater than 5.7 mg/liter; It has no skin irritation effect but with slight stimulus on eyes. It has no teratogenic, carcinogenic, and mutagenic effect on animals within experimental dose. For three generations of breeding experiments in rats, the non-effect dose is 0.12 mg/kg/day. For rats in two years, the non-effect dose in rats is 2 mg/kg/day. It is highly toxic to aquatic organisms. Trout-96 hours LD50:3.6 mg /l; bluegill sunfish-96 hours LC50: 9.6 micrograms/liter. It is also highly toxic to bees, oral-LD50: 0.009 g/head, contact-LD50: 0.002 g/head; However, the LT50 of its residue in the foliage is 4 hours. After 4 hours, the residue drug in the foliage agent is of low toxicity to bees. It has low toxicity to birds; quail, acute-oral LD50: greater than 2000 mg/kg, ducks, acute-oral LD50: 86.4 mg/kg.
For the preparation, rat acute-oral LD50: 650 mg/kg, rabbits acute-percutaneous LD50: greater than 2000 mg/kg. Rat acute-inhalation LD50: 1.1 mg/liter. It has irritation effect on eye and skin.
Its preparation is 1.8% Abamectin EC (18 g active ingredient contained per liter)
The Appearance of its preparation is light brown liquid. Its preparation can be stored stably at room temperature for more than 2 years.
Toxicity: According to Chinese pesticide toxicity grading standards, Abamectin belongs to highly toxic pesticides. For original drug; acute oral-rat LD50: l0 mg/kg; acute oral-mice LD50: 13 mg/kg; acute percutaneous administration-rabbits LD50: greater than 2000 mg/kg; acute percutaneous administration-rat LD50: greater than 380 mg/kg; acute inhalation-rat LC50 greater than 5.7 mg/liter; It has no skin irritation effect but with slight stimulus on eyes. It has no teratogenic, carcinogenic, and mutagenic effect on animals within experimental dose. For three generations of breeding experiments in rats, the non-effect dose is 0.12 mg/kg/day. For rats in two years, the non-effect dose in rats is 2 mg/kg/day. It is highly toxic to aquatic organisms. Trout-96 hours LD50:3.6 mg /l; bluegill sunfish-96 hours LC50: 9.6 micrograms/liter. It is also highly toxic to bees, oral-LD50: 0.009 g/head, contact-LD50: 0.002 g/head; However, the LT50 of its residue in the foliage is 4 hours. After 4 hours, the residue drug in the foliage agent is of low toxicity to bees. It has low toxicity to birds; quail, acute-oral LD50: greater than 2000 mg/kg, ducks, acute-oral LD50: 86.4 mg/kg.
For the preparation, rat acute-oral LD50: 650 mg/kg, rabbits acute-percutaneous LD50: greater than 2000 mg/kg. Rat acute-inhalation LD50: 1.1 mg/liter. It has irritation effect on eye and skin.
Its preparation is 1.8% Abamectin EC (18 g active ingredient contained per liter)
1. Abamectin has a slow insecticide, acaricide action with the number of dead insects reaching peak after three days. However, its application can immediately cause the feeding stop and spread of the pests.
2. Abamectin is highly toxic to fish, so avoid the contamination of rivers, ponds when spraying. Avoid spraying upon the foraging period of honeybees.
2. Abamectin is highly toxic to fish, so avoid the contamination of rivers, ponds when spraying. Avoid spraying upon the foraging period of honeybees.
Abamectin tablet
Specifications: 2mg/sheet
For the treatment of various kinds of nematodes, ticks, mites, fleas, lice and flies of horses, cattle, sheep, pigs, dogs, cats and other poultry both in vivo and in vitro.
Oral absorption for horse has a high efficacy (killing rate being 95% to 100%) on large round worm (common round worm, horse round worm, round worm without teeth), roundworm (Parascaris equorum), Enterobius (Oxyuris equi), stomach worms (Grand discreet Rasi nematodes, Hebronema nematodes), intestinal nematodes (trichostrongylus axei, Strongyloides), lung nematodes (dictyocaulus arnfieldi) and other adults worm and larvae).
Oral or subcutaneous administration of Abamectin for cattle, sheep has a high killing rate (97% to 100%) on Haemonchus, Ostertagia, Cooperia, Trichostrongyle (Trichostrongyle axei), round worm, Bunostomum, Nematodirus spp, Trichuris, Dictyocaulus, Chabertia ovina imago and 4-phase larva.
Oral administration or orally administration together with spice; the amount per time:
1. horses, cows, donkeys, mules, and sheep: take 1 tablet per 10kg of body weight.
2. pigs, fox, dogs, and cats: take 1 tablet per 6 kg of body weight; for treatment of Demodex canis, take one tablet per 3 kg of body weight; take continuously for five times at the interval of seven days.
3. chickens, ducks, geese, rabbits, and pigeons: take 1 tablet per 6~8 kg weight.
Specifications: 2mg/sheet
For the treatment of various kinds of nematodes, ticks, mites, fleas, lice and flies of horses, cattle, sheep, pigs, dogs, cats and other poultry both in vivo and in vitro.
Oral absorption for horse has a high efficacy (killing rate being 95% to 100%) on large round worm (common round worm, horse round worm, round worm without teeth), roundworm (Parascaris equorum), Enterobius (Oxyuris equi), stomach worms (Grand discreet Rasi nematodes, Hebronema nematodes), intestinal nematodes (trichostrongylus axei, Strongyloides), lung nematodes (dictyocaulus arnfieldi) and other adults worm and larvae).
Oral or subcutaneous administration of Abamectin for cattle, sheep has a high killing rate (97% to 100%) on Haemonchus, Ostertagia, Cooperia, Trichostrongyle (Trichostrongyle axei), round worm, Bunostomum, Nematodirus spp, Trichuris, Dictyocaulus, Chabertia ovina imago and 4-phase larva.
Oral administration or orally administration together with spice; the amount per time:
1. horses, cows, donkeys, mules, and sheep: take 1 tablet per 10kg of body weight.
2. pigs, fox, dogs, and cats: take 1 tablet per 6 kg of body weight; for treatment of Demodex canis, take one tablet per 3 kg of body weight; take continuously for five times at the interval of seven days.
3. chickens, ducks, geese, rabbits, and pigeons: take 1 tablet per 6~8 kg weight.
1. broad spectrum insecticide
The current reported insecticide spectrum of Abamectin contains 84 species. In China, it is mainly used for control of pests with small body, multiple generations and being prone to become drug-resistant such as pear psylla, and cotton aphid, leaf mining pests such as the Inter-American Blanchard, pest mites such as Tetranychus urticae, Calacarus carinatus Green, Tetranychus viennensis and pests with wide range of hosts and miscellaneous eating habits such as Plutella xylostella.
2. the unique mechanism of killing pests
Abamectin is a nerve toxic agent. Its mechanism is targeting to the GABAA receptor of insect neuron synapse or neuromuscular synapse, interfering with the information transfer of nerve endings, namely stimulating the nerve endings to release neurotransmitter inhibitor γ-aminobutyric acid (GA-BA), prompting the extensive opening of the GABA-gated chloride channel with chloride channel-activating effect. In this case, large influx of chloride ions cause nerve membrane potential being hyperpolarized, resulting in the inhibition of the nerve membrane, and thereby blocking the contact between nerve endings and muscle, thus causing insect paralysis, poor feeding, and death. Because of its unique mechanism of action, it has no cross-resistance with commonly-used agents. According to reports, in addition to GABA receptors controlled chloride channels, Abamectin can also affect other ligand-controlled chloride channels. For example, Ivermectin can induce the irreversible increase of membrane conduction of muscle fibers (non GABA innervations) of locust.
The current reported insecticide spectrum of Abamectin contains 84 species. In China, it is mainly used for control of pests with small body, multiple generations and being prone to become drug-resistant such as pear psylla, and cotton aphid, leaf mining pests such as the Inter-American Blanchard, pest mites such as Tetranychus urticae, Calacarus carinatus Green, Tetranychus viennensis and pests with wide range of hosts and miscellaneous eating habits such as Plutella xylostella.
2. the unique mechanism of killing pests
Abamectin is a nerve toxic agent. Its mechanism is targeting to the GABAA receptor of insect neuron synapse or neuromuscular synapse, interfering with the information transfer of nerve endings, namely stimulating the nerve endings to release neurotransmitter inhibitor γ-aminobutyric acid (GA-BA), prompting the extensive opening of the GABA-gated chloride channel with chloride channel-activating effect. In this case, large influx of chloride ions cause nerve membrane potential being hyperpolarized, resulting in the inhibition of the nerve membrane, and thereby blocking the contact between nerve endings and muscle, thus causing insect paralysis, poor feeding, and death. Because of its unique mechanism of action, it has no cross-resistance with commonly-used agents. According to reports, in addition to GABA receptors controlled chloride channels, Abamectin can also affect other ligand-controlled chloride channels. For example, Ivermectin can induce the irreversible increase of membrane conduction of muscle fibers (non GABA innervations) of locust.
Stratification flow activity refers to that: after its spray, Abamectin can penetrate into the leaf tissue and form a drug sac inside epidermis parenchyma cells for long-term storage, so Abamectin has good persistence. Because of its good stratification flow activity, Abamectin has good efficacy on killing pests such as pest mites, leaf miner flier, leaf miner moth and other borer pests or sucking insects that are difficult to be prevented and treated with common drugs. Abamectin is easily biodegradable in soil and water and can be absorbed by the soil without leaching and residue and do not pollute the environment; it also has no accumulation in vivo and no residual accumulation and persistence, and thus Abamectin should belong to pollution-free pesticides. Abamectin can also be broken down by the soil bacteria into derivatives having higher activity such as the insecticidal activity of plant nematodes.
There are many foreign studies regarding to the organism resistance and resistance mechanism on Abamectin. In 1980, Scott and Geoghiou had first discovered anti-pyrethroid indoor resistant-selection housefly strain (LPR) has 7.6-fold cross-resistance on abamectin. Further study showed that this phenomenon is due to the increased metabolism of multi-function oxidase (MFO) and reduced skin penetration with reduction of the skin penetration as the major resistance mechanism and is highly recessive inheritance. In 1991, Gampos and Dybas had discovered that two-spotted spider mites are resistant to abamectin with its resistance being also related to epidermis penetration and oxidative metabolism, and the development of the resistance of two-spotted spider mite to Abamectins is related to the duration of medication. Its resistance genetic is incomplete autosomal recessive inheritance. During the study on drug resistance selection of diamondback moth, Li Tengwu et al have found that genetic resistance to abamectin in Diamondback Moth is also incomplete autosomal recessive inheritance. Argentine and Clark has discovered the resistance of potato beetle on Abamectin with the mechanisms also being related to multi-function oxidase and carboxylesterase with its resistance heredity being similar to diamondback moth and T. urticae, namely also belonging to autosomal incompletely recessive inheritance. It has found that clover leaf miner, diamondback moth, and German cockroach also have certain resistance to abamectin.
CF mice acute-oral LD50: 13.6~23.8 mg/kg, CRCD mice: 10.6~11.3mg/kg, CRCD neonatal rats: 1.52mg/kg. The minimum dose for causing effect is: CR: CD neonatal rats daily: 0.12mg/kg, CRCD rats daily: 2.0mg/kg, Beagle dogs daily: 0.5mg/kg, monkeys daily: 2.0mg/kg. Rabbit, acute-percutaneous LD50> 2000mg/kg; Rats continuous administration: 8 weeks, the mice continuously administered: 94 weeks, non-effect daily dose: 4mg/kg, 2-year feeding rat, non-effect dose of 2mg/kg. Teratogenicity has showed that the non-effect dose of maternal toxicity in rats was 0.05mg/kg, and in mice was 1.6mg/kg. Ames test has showed that there was no genetic toxicity and no carcinogenic effects. Trout LC50: 3.2μg/L, carp LC50: 4.2μg/L, Daphnia LC50: 0.34 μg/L, diphtheria quail LD50: 2000mg/kg, ducks acute-oral LD50: 86.4mg/kg. Bees oral, LD50: 0.009μg; contact, LD50: 0.002μg/only.
It is a kind of 16-membered macrolide, farm-livestock dual antibiotics with strong insecticidal, acaricidal, nematicidal activity. It is of broad-spectrum, high efficiency and safety. It has strong stomach poisoning and contact-killing effect without being able to kill the eggs. Its mechanism of action is interfering with the neuro-physiological activity, affecting the transmission of cellular membrane chloride with GABA being the target site. When the drug stimulates the target sites, it can block the transmission process of motor nerve information, resulting in the signal of central nervous systems of pest being continuously received by motor neurons, causing rapid paralysis of pests within hours, poor feeding, and slow moving or not moving. Because they do not cause rapid dehydration of insect rapid dehydration, so the lethal effect is slow. They will generally die after 24d after. It is mainly used for the prevention and treatment of various kinds of pests such as diamondback moth, cabbage caterpillar, armyworm, and flea in vegetables or fruit trees, it is particularly efficient in treating insect pests resistant to other pesticides. The amount per hectare for treating vegetable pests is 10~20g with control efficiency of over 90%; for the control of citrus rust mite: 13.5~54g per hectare with residual time being as long as 4 weeks (reduce the dose to 13.5 to 27 g upon being mixed with mineral oil by which the residue time can be extended to 16 weeks); it can be used for control of carmine spider mite, tobacco budworm, bollworm and cotton aphid with good efficacy. In addition, it can also be used to control cattle parasitic diseases, such as Damalinia bovis, Boophilus microplus, and bovine foot mite. When used for the control of parasitic diseases, the dosage is 0.2mg/kg of body weight.
It has driving and killing effect on nematodes, insects and mites. It can be used for the treatment of nematodes disease, mite disease as well as parasitic disease of livestock and poultry.
It has good control efficacy and delayed resistance for various kinds of pests of citrus, vegetables, cotton, apples, tobacco, soybeans and tea.
It can be used for the prevention of many kinds of pests or pest mites of vegetables, fruit and cotton.
It has driving and killing effect on nematodes, insects and mites. It can be used for the treatment of nematodes disease, mite disease as well as parasitic disease of livestock and poultry.
It has good control efficacy and delayed resistance for various kinds of pests of citrus, vegetables, cotton, apples, tobacco, soybeans and tea.
It can be used for the prevention of many kinds of pests or pest mites of vegetables, fruit and cotton.
Abamectin is a colorless to yellowish crystalline powder. It is soluble in acetone, methanol,
toluene, chloroform, and ethanol, but insoluble in water. It is stable, and incompatible with
strong oxidizing agents. Abamectin is a mixture of Abamectins containing about 80%
Abamectin B1a and 20% Abamectin B1b. These two components, B1a and B1b, have very
similar biological and toxicological properties. The Abamectins are insecticidal/miticidal
compounds derived from the soil bacterium
Streptomyces avermitilis.
Abamectin is used to
control insect and mite pests of citrus, pear, and nut tree crops, and is used by homeown-
ers to control fi
re ants. It acts on the nervous system of insects, causing paralyzing effects.
Abamectin is a general use pesticide (GUP). It is grouped as toxicity class IV, meaning
practically non-toxic, requiring no precautionary statement on its label
Mixture of Abamectins, containing at least 80% of Abamectin B1a (C48H72O14) and not more than 20% of Abamectin B1b (C47H70O14). Used as acaricide, insecticide
Any of a group of broad spectrum antiparasitic antibiotics produced by the actinomycete, Streptomyces avermitilis.
1. The contents of a lyophilized tube of Streptomyces avermitilis MA-4680 is
transferred aseptically to a 250 ml Erlenmeyer flask containing 305 ml of
Medium 1: Dextrose 20 g, Peptone 5 g, Meat Extract 5 g, Primary Yeast 3 g,
NaCl 5 g, CaCO3 (after pH adjustment) 3 g, Distilled water 1000 ml, pH 7.0.
The inoculated flask is incubated for 3 days at 28°C on a rotary shaking
machine at a speed of 220 RPM in a 2 inch radius circular orbit. At the end of
this time, a 250 ml Erlenmeyer flask containing 50 ml of Medium 2 [Tomato
Paste 20 g, Modified Starch (CPC) 20 g, Primary Yeast 10 g, CoCl2·6H2O 0.005
g, Distilled water 1000 ml, pH 7.2-7.4] is inoculated with a 2 ml sample from
the first flask. This flask is incubated for 3 days at 28°C on a rotary shaking
machine at a speed of 220 RPM in a 2 inch diameter circular orbit. 50 Ml of
the resulting fermentation broth containing C-076 is effective against an
N.dubius infection in mice.
2. A lyophilized tube of Streptomyces avermitilis MA-4680 is opened aseptically and the contents suspended in 50 ml of Medium 1 in a 250 ml Erlenmeyer flask. This flask is shaken for 3 days at 28°C on a rotary shaking machine 220 RPM with a 2 inch diameter circular orbit. A 0.2 ml portion of this seed medium is used to inoculate a Slant of Medium 3: Dextrose 10.0 g , Bacto Asparagine 0.5 g, K2HPO4 40.5 g, Bacto Agar 15.0 g , Distilled water 1000 ml, pH 7.0. The inoculated slant medium is incubated at 28°C for 10 days and stored at 4°C until used to inoculate 4 more slants of Medium 3. These slants are incubated in the dark for 8 days. One of these slants is used to inoculate 3 baffled 250 ml Erlenmeyer flasks containing 50 ml of No. 4 Seed Medium: Soluble Starch 10.0 g, Ardamine 5.0 g, NZ Amine E 5.0 g, Beef Extract 3.0 g, MgSO4·7H2O 0.5 g, Cerelose 1.0 g, Na2HPO4 0.190 g, KH2PO4 182 g, CaCO3 0.5 g, Distilled water 1000 ml, pH 7.0-7.2. The seed flasks are shaken for 2 days at 27-28°C on a rotary shaking machine at 220 RPM with a 2 inch diameter circular orbit. The contents of these flasks are pooled and used to inoculate (5% inoculum) baffled 250 ml Erlenmeyer flasks containing 40 ml of various production media. Flasks containing media 2, 5 and 6 are incubated for 4 days at 28°C on a rotary shaking machine at 220 RPM with a 2 inch diameter circular orbit. The resulting broth containing C-076 is then harvested and tested for anthelmintic activity. In all cases 6.2 ml of whole broth and the solids obtained from centrifuging 25 ml of whole broth are fully active against N.dubius helminth infections in mice.
3. The one of the four slants of Medium 3 prepared as in Example 2 is used to inoculate a baffled 250 ml Erlenmeyer flask containing 50 ml of Seed Medium No. 4. The seed flask is shaken for 1 day at 27- 28°C on a rotary shaking machine at 220 RPM with a 2 inch diameter circular orbit. The seed flask is then stored stationary at 4°C until it is ready to be used. The contents of this flask are then used to inoculate (5% inoculum) 20 unbaffled 250 ml Erlenmeyer flasks containing 40 ml of Medium No. 2. After 4 days incubation at 28°C on a rotary shaking machine at 220 RPM with a 2 inch diameter circular orbit, 19 of the flasks are harvested and pooled. The combined fermentation broths containing C-076 are filtered affording 500 ml of filtrate and 84 g of mycelia. 78 G of mycelia are extracted with 150 ml of acetone for ? hour with stirring and the mixture filtered. The filter cake is washed with 50 ml of acetone and the filtrate and washings are combined and concentrated to 46.5 ml 30 Ml of the concentrate is adjusted to pH 4 with dilute hydrochloric acid and extracted 3 times with 30 ml portions of chloroform. The extracts are dried by filtering through dry Infusorial Earth (Super-Cel) combined and concentrated to dryness in vacuum. The oily residue of C-076 weighing 91.4 mg is dissolved in chloroform sufficient to make 3 ml of solution which represents 1% of broth volume. The C-076 (Abamectin) obtained in this recovery procedure is fully active against N.dubius infections in mice. In addition, the chloroform extraction achieved a 70 fold purification of C-076 from the whole broth.
2. A lyophilized tube of Streptomyces avermitilis MA-4680 is opened aseptically and the contents suspended in 50 ml of Medium 1 in a 250 ml Erlenmeyer flask. This flask is shaken for 3 days at 28°C on a rotary shaking machine 220 RPM with a 2 inch diameter circular orbit. A 0.2 ml portion of this seed medium is used to inoculate a Slant of Medium 3: Dextrose 10.0 g , Bacto Asparagine 0.5 g, K2HPO4 40.5 g, Bacto Agar 15.0 g , Distilled water 1000 ml, pH 7.0. The inoculated slant medium is incubated at 28°C for 10 days and stored at 4°C until used to inoculate 4 more slants of Medium 3. These slants are incubated in the dark for 8 days. One of these slants is used to inoculate 3 baffled 250 ml Erlenmeyer flasks containing 50 ml of No. 4 Seed Medium: Soluble Starch 10.0 g, Ardamine 5.0 g, NZ Amine E 5.0 g, Beef Extract 3.0 g, MgSO4·7H2O 0.5 g, Cerelose 1.0 g, Na2HPO4 0.190 g, KH2PO4 182 g, CaCO3 0.5 g, Distilled water 1000 ml, pH 7.0-7.2. The seed flasks are shaken for 2 days at 27-28°C on a rotary shaking machine at 220 RPM with a 2 inch diameter circular orbit. The contents of these flasks are pooled and used to inoculate (5% inoculum) baffled 250 ml Erlenmeyer flasks containing 40 ml of various production media. Flasks containing media 2, 5 and 6 are incubated for 4 days at 28°C on a rotary shaking machine at 220 RPM with a 2 inch diameter circular orbit. The resulting broth containing C-076 is then harvested and tested for anthelmintic activity. In all cases 6.2 ml of whole broth and the solids obtained from centrifuging 25 ml of whole broth are fully active against N.dubius helminth infections in mice.
3. The one of the four slants of Medium 3 prepared as in Example 2 is used to inoculate a baffled 250 ml Erlenmeyer flask containing 50 ml of Seed Medium No. 4. The seed flask is shaken for 1 day at 27- 28°C on a rotary shaking machine at 220 RPM with a 2 inch diameter circular orbit. The seed flask is then stored stationary at 4°C until it is ready to be used. The contents of this flask are then used to inoculate (5% inoculum) 20 unbaffled 250 ml Erlenmeyer flasks containing 40 ml of Medium No. 2. After 4 days incubation at 28°C on a rotary shaking machine at 220 RPM with a 2 inch diameter circular orbit, 19 of the flasks are harvested and pooled. The combined fermentation broths containing C-076 are filtered affording 500 ml of filtrate and 84 g of mycelia. 78 G of mycelia are extracted with 150 ml of acetone for ? hour with stirring and the mixture filtered. The filter cake is washed with 50 ml of acetone and the filtrate and washings are combined and concentrated to 46.5 ml 30 Ml of the concentrate is adjusted to pH 4 with dilute hydrochloric acid and extracted 3 times with 30 ml portions of chloroform. The extracts are dried by filtering through dry Infusorial Earth (Super-Cel) combined and concentrated to dryness in vacuum. The oily residue of C-076 weighing 91.4 mg is dissolved in chloroform sufficient to make 3 ml of solution which represents 1% of broth volume. The C-076 (Abamectin) obtained in this recovery procedure is fully active against N.dubius infections in mice. In addition, the chloroform extraction achieved a 70 fold purification of C-076 from the whole broth.
(Merck)Avomec [Veterinary] (Merial); Bovitin
[Veterinary] (Merial); Doratect [Veterinary] (Merial);
Duomectin [Veterinary] (Merial); Duotin [Veterinary]
(Merial); Endecto (Merck); Enzec (Merck); Enzek
(Merck); Parafoil (Merck); Vertimil (Zectin
(Merck).
Odorless off-white to yellow crystals from methanol. Does not hydrolyze in water at pH 3, 5, 7. Used as an acaricide and insecticide.
Abamectin is an insecticide and miticide. It is very toxic and causes adverse health effects
if swallowed and/or inhaled. Emulsifi
able concentrate formulations of Abamectin cause
slight to moderate eye irritation and mild skin irritation. The symptoms of poisoning
observed in laboratory animals include pupil dilation, vomiting, convulsions and/or trem-
ors, and coma. Abamectin acts on insects by interfering with the nervous system. At very
high doses, laboratory mammals develop symptoms of nervous system depression, inco-
ordination, tremors, lethargy, excitation, and pupil dilation. Very high doses have caused
death from respiratory failure in animals. Additionally, Abamectin has been reported to
cause reproductive effects. Abamectin blocks the nerval conduct system in insects, caus-
ing paralysis and death. Laboratory studies have indicated that abamectin may affect the
nervous system in experimental animals. A 1-year study with dogs given oral doses of
abamectin (0.5 and 1 mg/kg/day) caused adverse health effects, such as pupil dilation,
weight loss, lethargy, tremors, and recumbency.
Acaricide, Miticide, Insecticide, Anthelmentic: Used on fruit, vegetable and ornamental crops; pears,
citrus fruits, and nut crops; to control mite and insect pests,
and also to control household and lawn insects, including fire
ants. Approved by the EPA for use in ash trees for control of
emerald ash borer. A U.S. EPA restricted Use Pesticide (RUP).
ABACIDE®; AFFIRM®; AVID®,
AVID-EC®; AVOMEC®; DYNAMEC®; INJECT-A-
CIDE AV®; MK 936®(B 1A ); BOVITIN®; DORATECT®;
DUOMECTIN®; DUOTIN®; ENDECTO®; ENZEC®; L
676,863® (B 1A ); MK 0936®; MK 936®; PARAFOIL®;
VERTIMEC®, VERTIMIL®; VIVID®; ZECTIN®;
ZEPHEYR®; ZEPHYR®
Abamectin contains the closely related avermectin B1a and B1b as the
active ingredients. Avermectin B1a contains a sec-butyl moiety whereas
avermectin B1b contains an isopropyl moiety. Chemical degradation and
metabolism studies were conducted with avermectin B1a radiolabelled
with 3H or 14C at various positions of this large molecule. The overall fates
of avermectin B1a and B1b are similar since transformations at the butyl or
propyl moiety were not observed.
Avermectin B1a is stable to hydrolytic degradation, but it is readily degraded to numerous products in aqueous solutions, soil, glass and plant foliage/fruit surfaces after light irradiation. Isomerisation and O-demethylation appear to the primary degradation reactions. In addition, hydroxylation is a major metabolic reaction in animals. Significant amounts of the residues in plants and animals were characterised as unidentified polar components.
Avermectin B1a is stable to hydrolytic degradation, but it is readily degraded to numerous products in aqueous solutions, soil, glass and plant foliage/fruit surfaces after light irradiation. Isomerisation and O-demethylation appear to the primary degradation reactions. In addition, hydroxylation is a major metabolic reaction in animals. Significant amounts of the residues in plants and animals were characterised as unidentified polar components.
[3H]Avermectin B1a(1) was stable at 25 °C in pH 5,7 and 9 solutions up to
28 days. Less than 5% degradation was observed (PSD, 1992).
[14C]Avermectin B1a degraded rapidly in aqueous solution when exposed to sunlight with an observed DT50 of 3.5-12 hours. The △8,9 geometric isomer of avermectin B1a (2) was tentatively identified in aqueous solution (containing 2% acetone as photosensitiser) after irradiation under artificial light. Crouch et al. (1991) reported the photodegradation of avermectin B1a, as a thin film on glass surfaces under artificial light (>260 nm). At least 10 photodegradation products were characterised including the geometric isomers (2, 3), hydroxylated (4-7) and O-demethylated products (8) shown in Scheme 1.
[14C]Avermectin B1a degraded rapidly in aqueous solution when exposed to sunlight with an observed DT50 of 3.5-12 hours. The △8,9 geometric isomer of avermectin B1a (2) was tentatively identified in aqueous solution (containing 2% acetone as photosensitiser) after irradiation under artificial light. Crouch et al. (1991) reported the photodegradation of avermectin B1a, as a thin film on glass surfaces under artificial light (>260 nm). At least 10 photodegradation products were characterised including the geometric isomers (2, 3), hydroxylated (4-7) and O-demethylated products (8) shown in Scheme 1.
Preparation Products And Raw materials
Preparation Products
Abamectin Supplier
Tel +8619931165850
Email hbjbtech@163.com
Products Intro Product Name:Abamectin
CAS:71751-41-2
Purity:99.99% Package:1kg;1USD
CAS:71751-41-2
Purity:99.99% Package:1kg;1USD
Tel +8615398038360
Email lionel@accenturebio.com
Products Intro Product Name:Abamectin
CAS:71751-41-2
Purity:99% Package:1KG;10KG;25KG
CAS:71751-41-2
Purity:99% Package:1KG;10KG;25KG
Tel +8613288715578
Email sales@hbmojin.com
Products Intro Product Name:Abamectin
CAS:71751-41-2
Purity:99% Package:25KG;USD
CAS:71751-41-2
Purity:99% Package:25KG;USD
Tel +8618239973690
Email sales@suikangpharm.com
Products Intro Product Name:Abamectin
CAS:71751-41-2
Purity:≥92% Package:1kg;0.00;USD
CAS:71751-41-2
Purity:≥92% Package:1kg;0.00;USD
Tel +86-17331933971 +86-17331933971
Email deasea125996@gmail.com
Products Intro Product Name:Abamectin
CAS:71751-41-2
Purity:99.99% Package:1kg;10USD
CAS:71751-41-2
Purity:99.99% Package:1kg;10USD
Abamectin manufacturers
Related articles
The harmful of abamectin to humansFeb 19,2024
Where is Abamectin usedNov 27,2019
Related Product Information
- CARVEOL
- (4''r)-4''-(acetylamino)-4''-deoxy-avermectin b1solution,Avermectin B1, 4-(acetylamino)-4-deoxy-, (4R)-,(4''R)-4''-(ACETYLAMINO)-4''-DEOXY-AVERMECTIN B1
- Methylamino abamectin
- Tetrahydropyranyl-4-acetic acid
- ETHYL 2-METHYL-3-PENTENOATE
1of4
PROMPT×
PROMPT
The What'sApp is temporarily not supported in mainland China
The What'sApp is temporarily not supported in mainland China
Cancel
Determine