Melamine-formaldehyde resin (MFR) is an active
ingredient of strong (reinforced) plasters. Sensitization
was reported in a plaster-room technician, who applied
resin-reinforced pIaster casts, and in dental technicians.
MFR was contained in a strong dental pIaster
used for mouldings. Used as a textile finish res in, it was
also found to be an allergen in a women who replaced
clothes in a store. MFR also releases formaldehyde,
which may be the sensitizer.
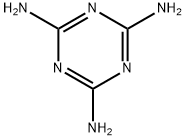
Melamine is a white solid organic compound whose molecules consist of a sixmembered heterocyclic ring of alternate carbon and nitrogen atoms with three amino groups attached to the carbons. Condensation polymerization with methanal or other aldehydes produces melamine resins, which are important thermosetting plastics.
It is used to make high-pressure laminating resins
(e.g., decorative countertops), molded compounds (e.g.,
dinnerware), and surface coating resins (e.g., appliance
finishes and automotive topcoats). Additional major products
are textile and paper treatment resins. Miscellaneous uses
include adhesive resins for gluing lumber, plywood, and
flooring, and resins for leather tanning agents. Melamine,
melamine cyanurate, other melamine salts, and guanidine
compounds are currently the most used group of nitrogencontaining
flame retardants. Melamine is used as a flame
retardant additive for polypropylene and polyethylene.
Melamine cyanurate is employed commercially as a flame
retardant for polyamides and terephthalates.
Forms synthetic resins with formaldehyde.
The compound now is synthesized from urea.
melamine: A white crystalline compound,C3N6H6. Melamine is a cycliccompound having a six-memberedring of alternating C and N atoms,with three NH2 groups. It can becopolymerized with methanal to givethermosetting melamine resins,which are used particularly for laminatedcoatings.
Melamine is prepared almost exclusively by the urea
process—the action of ammonia on urea. It is produced
worldwide.
The standard route to melamine is from urea. Urea is
heated in the presence of ammonia at 250-350??C and 4--20 MPa. The
reaction probably involves the simultaneous dehydration and hydration of
urea to form cyanamide and ammonium carbamate; trimerization of the
cyanamide then leads to melamine:
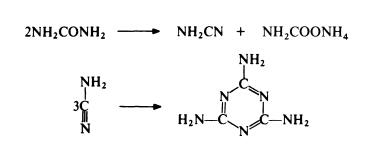
Thus only 50% of the urea used gives melamine in one step and ammonium
carbamate has to be separated and converted to urea for recycling. Despite
this limitation, the urea route is the most economical of currently available
routes.
ChEBI: Melamine is a trimer of cyanamide, with a 1,3,5-triazine skeleton. It has a role as a xenobiotic metabolite. It is functionally related to a cyanamide. It is a conjugate base of a melamine(1+).
Colorless to white monoclinic crystals or prisms or white powder. Sublimes when gently heated.
Melamine is incompatible with strong oxidizing agents and strong acids . Neutralizes acids in exothermic reactions to form salts plus water. May be incompatible with isocyanates, halogenated organics, peroxides, phenols (acidic), epoxides, anhydrides, and acid halides. Flammable gaseous hydrogen may be generated in combination with strong reducing agents, such as hydrides.
Toxic by ingestion, skin, and eye irritant.
Questionable carcinogen.
Literature sources indicate that Melamine is nonflammable.
Melamine-formaldehyde resin (MFR) results from condensation of melamine and formaldehyde. It is anactive ingredient of strong (reinforced) plasters, such as industrial or some dental plasters used for molding.It is also used as a textile finish resin. MFR acts as an allergen generally because of formaldehyde releasing (see Chap. 40)
Moderately toxic by
ingestion and intraperitoneal routes. An eye,
skin, and mucous membrane irritant. Causes
dermatitis in humans. Questionable
carcinogen with experimental carcinogenic
and tumorigenic data. Experimental
reproductive effects. Mutation data
reported. When heated to decomposition it
emits toxic fumes of NOx and CN-.
Manufactured from urea, melamine
is used in the manufacture of plastics, melamineformaldehyde resins; rubber, synthetic textiles; laminates,
adhesives, and molding compound
If this chemical gets into the eyes, remove any contact lenses at once and irrigate immediately for at least 15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts the skin, remove contaminated clothing and wash immediately with soap and water. Seek medical attention immediately. If this chemical has been inhaled, remove from exposure, begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR if heart action has stopped. Transfer promptly to a medical facility. When this chemical has been swallowed, get medical attention. Give large quantities of water and induce vomiting. Do not make an unconscious person vomit.
A bioassay of melamine was
conducted in rats and mice by NTP. Male F344 rats and
B6C3F1 mice were administered melamine in their diets at
concentrations of 2250 or 4500 ppm daily for 103 weeks.Female rats were fed 4500 or 9000 ppm melamine. At the end
of 111 weeks, surviving animals were killed and examined.
Color Code—Blue: Health Hazard/Poison: Store in a secure poison location. Prior to working with this chemical you should be trained on its proper handling and storage. Store in tightly closed containers in a cool, well-ventilated area away from strong oxidizers and strong acids. Where possible, automatically pump liquid from drums or other storage containers to process containers. A regulated, marked area should be established where this chemical is handled, used, or stored in compliance with OSHA Standard 1910.1045.
Crystallise Melamine from water or dilute aqueous NaOH. It sublimes at ~240o on prolonged heating. [Beilstein 26 I 74, 26 II 132, 26 III/IV 1253.]
Incompatible with oxidizers (chlorates,
nitrates, peroxides, permanganates, perchlorates, chlorine,
bromine, fluorine, etc.); contact may cause fires or explosions. Keep away from alkaline materials, strong bases,
strong acids, oxoacids, epoxides. Melamine neutralizes
acids in exothermic reactions to form salts plus water. May
be incompatible with isocyanates, halogenated organics,
peroxides, phenols (acidic), epoxides, anhydrides, and acid
halides. Flammable gaseous hydrogen may be generated in
combination with strong reducing agents such as hydrides,
nitrides, alkali metals, and sulfides.