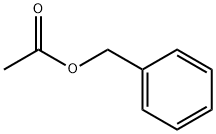
Benzyl acetate has a characteristic flowery (jasmine) odor and a bitter, pungent taste. It is the main component of jasmine absolute and gardenia oils. It occurs as a minor component in a large number of other essential oils and extracts. It is a colorless liquid with a strong, fruity, jasmine odor. In terms of volume, benzyl acetate is one of the most important fragrance and flavor chemicals. Although benzyl acetate is present in some essential oils at levels up to 65%, most of the commercial product is of synthetic origin.
Benzyl acetate is a colorless liquid with a fruity odor. On burning and decomposition, it produces irritating fumes. Benzyl acetate reacts with strong oxidants causing fire and explosion hazard.

Benzyl acetate is stable under normal conditions of use. Heating to decomposition may release carbon monoxide, carbon dioxide and other potentially toxic fumes and gases. Vapors may form explosive mixtures with air at elevated temperatures (> 90°C / 194°F). Avoid heat, open flames and other potential sources of ignition.
It has been used as a food additive in fruit flavours and as a component of perfumes since the early 1990s and is widely used as a fragrance in soaps, detergents and incense. There is widespread human exposure to benzyl acetate by ingestion, skin application and inhalation.
Present as a main constituent in several oils and flower absolutes: ylang-ylang, cananga, neroli, jasmine, hyacinth,
gardenia, tuberose. It has been isolated from the essential oil of the flowers of Loiseleuria procumbens Desv. (azelea). Also
reported found in apricot, cooked asparagus, mozzarella cheese, grilled beef, cooked pork, malt whiskey, fresh mango, malt, wort
and clams.
Benzyl acetate is used as an artificial jasmine and other
perfumes, soap perfume, flavoring agent, solvent for cellulose
acetate and nitrate, natural and synthetic resins, oils,
lacquers, polishes, printing inks, and varnish removers.
Benzyl acetate can be used as a high boiling point solvent in coatings, like ink coating, binding agent and paint remover. It is used in soap and other chemical essences and has the effect of promote in floral and fantasy essences in jasmine, white orchid, fragrant plantain lily, gekkakou, narcissus and other essences. It is a plasticizer for ionophore membranes.
Benzyl acetate is produced by reacting benzyl chloride with an alkali
acetate or by esterifying benzyl alcohol with
acetic anhydride. It is also formed in the oxidation of toluene in the presence of acetic acid or
acetic anhydride. Benzyl acetate is one of the
most used odorants. It is also used as a flavor and,
to a small extent, as a high-boiling solvent.
ChEBI: Benzyl acetate is the acetate ester of benzyl alcohol. It has a role as a metabolite. It is an acetate ester and a benzyl ester.
Benzyl acetate is produced by the interaction of benzyl chloride and sodiumacetate, by acetylation of benzyl alcohol, or from benzaldehyde and acetic acid with zinc dust.
Taste characteristics at 40 ppm: sweet and fruity
Colorless liquid with an odor of pears.
Benzyl acetate is an ester. Esters react with acids to liberate heat along with alcohols and acids. Strong oxidizing acids may cause a vigorous reaction that is sufficiently exothermic to ignite the reaction products. Heat is also generated by the interaction of esters with caustic solutions. Flammable hydrogen is generated by mixing esters with alkali metals and hydrides. Benzyl acetate is incompatible with strong oxidizing agents. Benzyl acetate is also incompatible with acids, bases and reducing agents.
A poison by inhalation. Moderately toxic
by ingestion. Combustible. Upper respiratory tract
irritant. Questionable carcinogen.
Harmful if inhaled. May be harmful if swallowed or absorbed through the skin. Vapor or mist is irritating to the eyes, mucous membrane and upper respiratory tract.
Exposures to benzyl acetate cause adverse health effects. The symptoms of toxicity and poisoning include irritation to the skin, eyes, burning sensation, confusion, dizziness, drowsiness, labored breathing, sore throat, nausea, vomiting, and diarrhea. Benzyl acetate also causes
adverse health effects to the respiratory tract and the CNS system with neurological effects.
Benzyl acetate is combustible.
Flammability and Explosibility
Non flammable
A poison by inhalation.Moderately toxic by ingestion and subcutaneous routes.Human systemic effects by inhalation: an antipsychotic,unspecified respiratory and urinary system effects.Questionable carcinogen with experimental tumorigenicdata. Combusti
Not listed by ACGIH, IARC,
NTP, or California Proposition 65.
The esters of benzyl alcohol, such as the acetate, benzoate, cinnamate and hydrocinnamate, are rapidly hydrolysed in vivo to benzyl alcohol which is then oxidized to benzoic acid and excreted as hippuric acid
Benzyl acetate should be kept stored in a cool, dry place with the container closed when
not in use.
Purify the acetate by fractional distillation, preferably in a good vacuum. Values of n25 of 1.5232-1.5242 are too high and should be nearer to 1.4994. [Merker & Scott J Org Chem 26 5180 1961, Beilstein 6 IV 2262.]
Exposures to benzyl acetate far above the OEL may result in unconsciousness. After
handling and using benzyl acetate, workers should wash thoroughly and remove contaminated clothing, washing it before reuse. Workers should avoid any kind of contact of
benzyl acetate with the eyes, skin, ingestion, and inhalation. Workers should wear safety
glasses and chemical goggles to avoid splashing of the chemical substance during work,
and wear appropriate protective gloves and clothing to prevent skin exposure