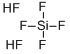Hexafluorosilicic acid is a kind of inorganic acid. It is majorly used for the fluoridation of water in United State to minimize the incidence of dental caries and dental fluorosis. For chemical synthesis, it is majorly used for the manufacturing of aluminum fluoride and cryolite as well as many kinds of hexafluorosilicate salts. It can also be used for the production of silicon and silicon dioxide. It can also be used as an electrolyte in the Betts electrolytic process for refining lead. It is also a specialized reagent in organic synthesis for cleaving Si–O bonds of silyl ethers.
Fluosilicic acid,H2SiF6, also known as hydrofluorosilicic acid,is a colorless liquid that is soluble in water. It is highly corrosive and toxic,attacking glass and stoneware. Fluosilicic acid is used in water fluoridation, electroplating, and in manufacturing enamels and cement.
Fluorosilicic acid is a transparent, colorless
fuming liquid.
d 1.220 g cm?3 for a 25% aq solution.
A fluoride source with both protic and Lewis acid properties providing efficient cleavage of silicon–oxygen bonds, e.g. silyl ether
deprotection.
Hexafluorosilicic acid is commonly used as a source of fluoride. It is converted to a variety of useful hexafluorosilicate salts. It is also used as an electrolyte in the Betts electrolytic process for refining lead. It is an important organic reagent for cleaving Si-O bonds of silyl ethers. Further, it is used as wood a preservation agent and also used in surface modification of calcium carbonate.
A 1-2% solution is used widely for sterilizing equipment in brewing and bottling establishments. Other concentrations are used in the electrolytic refining of lead, in electroplating, for hardening cement, crumbling lime or brick work, for the removal of lime from hides during the tanning process, to remove molds, as preservative for timber.
A colorless fuming liquid with a penetrating pungent odor. Corrosive to metals and tissue. Both the fumes and very short contact with the liquid can cause severe and painful burns. Used in water fluoridation, in hardening cement and ceramics, as a wood preservative.
Fumes in air. Soluble in water with release of heat and corrosive fumes.
Hexafluorosilicic acid can react with strong acids (such as sulfuric acid) to release fumes of toxic hydrogen fluoride. Attacks glass and materials containing silica. Reacts exothermically with chemical bases (examples: amines, amides, inorganic hydroxides). Reacts with active metals, including iron and aluminum to dissolve the metal and liberate hydrogen and/or toxic gases. Can initiate polymerization in certain alkenes. Reacts with cyanide salts and compounds to release gaseous hydrogen cyanide. Flammable and/or toxic gases are also often generated by reactions with dithiocarbamates, isocyanates, mercaptans, nitrides, nitriles, sulfides, and weak or strong reducing agents. Additional gas-generating reactions may occur with sulfites, nitrites, thiosulfates (to give H2S and SO3), dithionites (SO2), and carbonates. Can catalyze (increase the rate of) chemical reactions. Decomposes when heated to the boiling point to produce very toxic and corrosive hydrogen fluoride gas.
Extremely corrosive by skin contact and
inhalation.
Inhalation of vapor produces severe corrosive effect on mucous membrane. Ingestion causes severe burns of mouth and stomach. Contact with liquid or vapor causes severe burns of eyes and skin.
Special Hazards of Combustion Products: Irritating fumes of hydrogen fluoride may form in fire.
Flammability and Explosibility
Not classified
Hydrofluorosilicic acid (H2SiF6) is a colorless to light brown liquid. It is also manufactured
from calcium fluoride or other fluoride-containing products. Hydrofluorosilic acid
is a strong depressant for many silicates during flotation of a number of oxidic minerals.
It is used for gangue depression during flotation of tin, columbite and tantalite.
Poison by subcutaneous route. A corrosive irritant to sktn, eyes, and mucous membranes. Will react with water or steam to produce toxic and corrosive fumes. When heated to decomposition it emits toxic fumes of F-. See also FLUORIDES.
A solution of fluorosilicic acid is used
for sterilization in the brewing and bottling industry, elec trolytic refining of lead; electroplating, hardening cement;
removing mold, and others.
this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at leastFluosilicic acid 134515 min, occasionally lifting upper and lower lids. Seekmedical attention immediately. If this chemical contactsthe skin, remove contaminated clothing and wash immediately with soap and water. Seek medical attention immediately. If this chemical has been inhaled, remove fromexposure, begin rescue breathing (using universal precautions, including resuscitation mask) if breathing hasstopped and CPR if heart action has stopped. Transferpromptly to a medical facility. When this chemical hasbeen swallowed, get medical attention. If victim is conscious, administer water or milk. Do not induce vomiting.Medical observation is recommended for 24-48 h afterbreathing overexposure, as pulmonary edema may bedelayed. As first aid for pulmonary edema, a doctor orauthorized paramedic may consider administering a corticosteroid spray.Note to physician: Inject intravenously 10 mL of 10% calcium gluconate solution. Gastric lavage with lime water of1% calcium chloride.
(1) Color Code—White: Corrosive or ContactHazard; Store separately in a corrosion-resistant location.(2) Color Code—Blue: Health Hazard/Poison: Store in asecure poison location. Prior to working with this chemicalyou should be trained on its proper handling and storage.Store in a cool, dry area that is well ventilated. Protect fromdamage. Avoid acids. Concentrated solution can be storedin glass but lead is preferred.
UN1778 Fluorosilicic acid, Hazard class: 8;
Labels: 8-Corrosive material.
The aqueous solution is a strong acid.
Reacts with water or steam to produce toxic and corrosive
fumes of hydrogen fluoride. Incompatible, and may react violently with: bases, aliphatic amines; alkanolamines,
alkylene oxides; aromatic amines; amides, ammonia,
ammonium hydroxide; calcium oxide; epichlorohydrin, iso cyanates, oleum, organic anhydrides; sulfuric acid; strong
oxidizers; vinyl acetate; water. Attacks glass, concrete, and
ceramics. The anhydrous form dissociates almost instantly
into silicon tetrafluoride and hydrogen fluoride.
Add slowly to a large amount
of soda ash in solution. Discharge to sewer with large
volumes of water
Robinson, Tim. "Innovative Processes in Electrometallurgy." Innovative Process Development in Metallurgical Industry. Springer International Publishing, 2016. 385- 392.
Sarawade, Pradip B., et al. "Recovery of high surface area mesoporous silica from waste hexafluorosilicic acid (H 2 SiF 6) of fertilizer industry." Journal of hazardous materials 173.1 (2010): 576-580.
Kauffman, Joel M. "Water fluoridation: a review of recent research and actions." Journal of American Physicians and Surgeons 10.2 (2005): 38.
Krot, V. V., et al. "ChemInform Abstract: Preparation of Amorphous Silicon Dioxide from Hexafluorosilicic Acid." Cheminform 23.48(1992):no-no.
Zorya, L., and V. Krot. "Method of high-purity silica production from hexafluorosilicic acid." Reaction Kinetics & Catalysis Letters 50.1-2(1993):349-354.