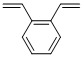
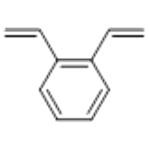
Divinylbenzene is a clear yellow liquid with an aromatic odour. It is a highly flammable and reactive chemical, and the liquid and vapour are combustible. It is incompatible with strong acids and oxidising agents and peroxides. Divinylbenzene reacts vigorously with strong oxidising agents. It also reacts exothermically with both acids and bases, with reducing agents (such as alkali metals and hydrides), and release gaseous hydrogen. May react exothermically with both acids and bases or initiators undergo exothermic polymerisation. It is an extremely versatile chemical cross-linking agent used to improve polymer properties. Divinylbenzene has extensive industrial applications for example, in the manufacture of adhesives, plastics, elastomers, ceramics, biological materials, catalysts, membranes, pharmaceuticals, specialty polymers, and ion exchange resins, as coatings, divinyl benzene has been reported to improve surface properties of rubber goods for biomedical applications, used in photocurable protective coatings for wood, metal, glass, and plastic materials.
Divinylbenzene is a water-white to straw colored liquid. Slightly less dense than water and insoluble in water. Vapors may be toxic. Used in making rubber.Synthetic rubber is any one of a group of man-made elastomers that approximate one or more of the properties of natural rubber(polyisoprene). Divinylbenzene has been used in Styrene-Butadiene copolymers (SBR) as adhesives and as processing aids in the extrusion of general purpose rubbers. Rubbers used as impact modifiers such as Methacrylate-Butadiene-Styrene (MBS) have been improved with the inclusion of Divinylbenzene.
Divinylbenzene (DVB) is an extremelyversatile cross-linking agent that also improves polymer properties. it has been used to manufacture adhesives,plastics, elastomers, ceramics, biologicalmaterials, coatings, catalysts, membranes, pharmaceuticals, specialty polymers, and ion exchange resins.
DVB has been found to improve the hightemperature strength of adhesives used tobond aluminum to aluminum. Used in anacrylic hydrosol to bond Bakelite resin andpolyester film, DVB was found to increasecreep rupture time dramatically. DVB hasfound utility in cross-linked styrene/butadienepolymers used for caulks and mastics for theconstruction industry.
DVB has been used in the production ofreversed-phase packings for liquidchromatography. Highly cross-linkedpolystyrene packings have been shown tofunction under extreme conditions of highpressures and temperatures. Ion exchangechromatography (IEC) is applicable to theseparation of almost any type of chargedmolecule, from large proteins to smallnucleotides and amino acids.
- F. C. Huang, C. H. Ke, C. Y. Kao, W. C. Lee, Preparation and application of partially porous poly(styrene‐divinylbenzene) particles for lipase immobilization, Journal of Applied Polymer Science, 2001, vol. 80, pp. 39-46
- Teresa Norber-King, Toxicity Reduction and Toxicity Identification Evaluation for effluents, ambient waters, and other aqueous media, 2001, ISBN 1-880611-64-3
DVB exists as o-, m-, and p-isomers. The commercial product contains all 3 isomers, but m-isomer predominates. Usually contains an inhibitor to preventpolymerization. The CAS number for the mixed isomersappears on most regulatory lists.DVB is a pale, straw-colored liquid. Molecularweight= 130.20; Specific gravity (H2O:1) 5 0.93; Boilingpoint= 195-200℃; Freezing/Melting point = 2 66.7to 252℃; Vapor pressure= 0.7 mmHg at 20℃; Flashpoint= 62℃ (cc); 76℃ (oc); Autoignition temperature=469℃. Explosive limits: LEL = 0.7%; UEL= 6.2%.Hazard Identification (based on NFPA-704 M RatingSystem): Health 1, Flammability 2, Reactivity 2. Insolublein water.
A water-white to straw colored liquid. Slightly less dense than water and insoluble in water. Flash point below 141°F. Vapors may be toxic. Used in making rubber.
Comonomer for preparation of crosslinked
polymers in production of ion exchange
beads and gel permeation chromatography
polystyrene beads; polymerization monomer
for synthetic rubber, drying oils, and casting
resins
Divinylbenzene is used as a reactive monomer in polyester resins and for the synthesis of highly crosslinked macroporous styrene as well as a crosslinking agent. It acts as an intermediate in the production of ion exchange resin. Further, it is used in making rubber, petroleum production and polymeric core shell nanoparticles. In addition, it is used to synthesize monodispersed poly divinylbenzene microspheres.
Divinylbenzene is a specialty monomer used primarily to make cross-linked polystyrene resins. The largest use of divinylbenzene (DVB) [CAS: 1321-74-0] is in ion-exchange resins for domestic and industrial water softening. Ion-exchange resins are also used as solid acid catalysts for certain reactions, such as esterification. Divinylbenzene is manufactured by dehydrogenation of diethylbenzene, which is an internal product in the alkylation plant for ethylbenzene production.
ChEBI: 1,2-Divinylbenzene is a member of styrenes.
Divinylbenzene (DVB) consists of a combination of meta and para isomers of DVB and ethylvinylbenzene. It can be used as a monomer for the preparation of linear polydivinylbenzene by anionic polymerization.
Flammable. Insoluble in water.
Divinylbenzene may react vigorously with strong oxidizing agents. Can react exothermically with reducing agents (such as alkali metals and hydrides) to release gaseous hydrogen. May react exothermically with both acids and bases. May in the presence of various catalysts (such as acids) or initiators undergo exothermic polymerization. Inhibited by presence of an additive. When uninhibited violent polymerization may occur . Substitution at the benzene nucleus occurs by halogenation (acid catalyst), nitration, sulfonation, and the Friedel-Crafts reaction.
Divinyl benzene is an irritant of
eyes, nose, and mucous membranes.
Mild respiratory irritation occurred in
workers exposed to 0.4–4ppm divinyl
benzene. Mild irritation was also reported
from skin and eye contact.
Divinylbenzene is combustible.
Flammability and Explosibility
Not classified
Mildly toxic by ingestion.
An eye irritant. Combustible. When heated to
decomposition it emits acrid smoke and irritating
fumes.
Tumorigen,Mutagen. This compound is used as a monomer for thepreparation of special synthetic rubbers, drying oils, ionexchange resins and casting resins, and in polyester resinmanufacture
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit.
Divinyl benzene was weakly genotoxic in
vivo, inducing a dose-dependent increase in
sister chromatid exchanges and an increase
in the frequency of chromosome aberrations in
male mice exposed at concentrations of up to
75ppm for 3 days.
The 2003 ACGIH threshold limit valuetime-
weighted average (TLV-TWA) for divinyl
benzene is 10ppm (53mg/m3).
Color Code—Red: Flammability Hazard: Store ina flammable liquid storage area or approved cabinet awayfrom ignition sources and corrosive and reactive materials.Prior to working with DVB you should be trained on itsproper handling and storage. Before entering confined spacewhere this chemical may be present, check to make sure thatan explosive concentration does not exist. Store in tightlyclosed containers in a cool, well-ventilated area away frommetallic salts (such as ferric and aluminum chlorides) andoxidizers (such as perchlorates, peroxides, permanganates,chlorates, and nitrates). Store in tightly closed containers ina cool, well-ventilated area away from heat. Sources of ignition, such as smoking and open flames, are prohibited wherethis chemical is used, handled, or stored in a manner thatcould create a potential fire or explosion hazard. Metal containers involving the transfer of 5 gallons or more of thischemical should be grounded and bonded. Drums must beequipped with self-closing valves, pressure vacuum bungs,and flame arresters. Use only nonsparking tools and equipment, especially when opening and closing containers ofthis chemical.
Diethylbenzene requires a shipping label of“FLAMMABLE LIQUID.” It falls in Hazard Class 3 andPacking Group II
Purify divinylbenzene by dissolving in Et2O, shaking with H2O, drying over CaCl2, filtering, evaporating and distilling in vacuo. It polymerises within 2-3days unless 4-tertbutylcatechol (0.05%) is added as stabilizer. [Fries & Bestian Chem Ber 69 715 1936, Beilstein 5 III 1366.]
Forms explosive mixture with air.Violent reaction with strong oxidizers, metallic salts. Ableto polymerize; add inhibitor and monitor to insure effectivelevels are maintained at all times. May accumulate staticelectrical charges, and may cause ignition of its vapors.