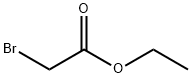
Ethyl bromoacetate is a clear, colorless tolight-yellow liquid. Molecular weight = 167.02; Boilingpoint = 159℃. Freezing point ≤-20℃; Flashpoint = 48℃. Hazard Identification (based on NFPA-704 MRating System): Health 2, Flammability 2, Reactivity 0.Insoluble in water
Ethyl bromoacetate is a clear, colorless to light-yellow liquid and has a pungent odor. It is miscible with ethanol, ether, benzene and with other oxygenated and aromatic solvents, insoluble in water and partially decomposed by water.
Ethyl bromoacetate is widely used as an alkylating reagent involved in the Reformatsky reaction to prepare the beta-hydroxy esters by reacting with carbonyl compounds. It is also used for the syntheses of witting reagent, artificial diethylstilbestrol antigen, 3-phenyl-1-naphthol and steroidal thiazolidinone derivatives. It finds applications in the preparation of reversibly photoresponsive coumarin-stabilized polymeric nanoparticles in an aqueous medium which act as a detectable drug carrier.
Ethyl Bromoacetate is used as a synthetic organic chemical intermediate as well as a pharmaceutical and agricultural intermediate. It is used in the synthesis of metabolites of carcinogenic PAHs. It is also used in the preparation of steroidal antiestrogens through cyclic condensation. Ethyl Bromoacetate is a reactant in the preparation of antim icrobial and antioxidant coumarinyloxymethyl-thiadiazolone.
Ethyl bromoacetate is commonly synthesized by sulfuric acid catalyzed esterification of monobromoacetic acid (MBAA). After reaction completion, excess acid is washed out and the product purified via distillation under reduced pressure if necessary (Stenger, 1978; Korhonen, 1984).
Ethyl bromoacetate on derivatisation reaction with p-t-butyl calix[4]arene yields 1,3-diester substituted calix[4]arene. It also undergoes Suzuki type cross-coupling reactions with arylboronic acids cocatalyzed by copper(I) oxide.
Ethyl bromoacetate appears as a clear, colorless liquid. A lachrymator. Toxic by ingestion, inhalation and skin absorption; a strong irritant of the skin. Insoluble in water and soluble in alcohol, benzene, and ether. Specific gravity of 1.5.
Flammable. Insoluble in water.
Ethyl bromoacetate is a halogenated ester. Esters react with acids to liberate heat along with alcohols and acids. Strong oxidizing acids may cause a vigorous reaction that is sufficiently exothermic to ignite the reaction products. Heat is also generated by the interaction of esters with caustic solutions. Flammable hydrogen is generated by mixing esters with alkali metals and hydrides.
Toxic by ingestion, inhalation, and skin
absorption; strong irritant.
TOXIC; inhalation, ingestion or contact (skin, eyes) with vapors, dusts or substance may cause severe injury, burns or death. Bromoacetates and chloroacetates are extremely irritating/lachrymators. Reaction with water or moist air will release toxic, corrosive or flammable gases. Reaction with water may generate much heat that will increase the concentration of fumes in the air. Fire will produce irritating, corrosive and/or toxic gases. Runoff from fire control or dilution water may be corrosive and/or toxic and cause pollution.
HIGHLY FLAMMABLE: Will be easily ignited by heat, sparks or flames. Vapors form explosive mixtures with air: indoors, outdoors and sewers explosion hazards. Most vapors are heavier than air. They will spread along ground and collect in low or confined areas (sewers, basements, tanks). Vapors may travel to source of ignition and flash back. Substance will react with water (some violently) releasing flammable, toxic or corrosive gases and runoff. Contact with metals may evolve flammable hydrogen gas. Containers may explode when heated or if contaminated with water.
A poison. An irritant to
skin, eyes, and mucous membranes.
Questionable carcinogen with experimental
neoplastigenic data. Flammable liquid when
exposed to heat, flame, and oxidizers. Will
react with water or steam to produce toxic
and corrosive fumes. To fight fire, use water
as a fire blanket. When heated to
decomposition or on contact with acid or
acid fumes, it emits highly toxic fumes of
Br-. See also BROMIDES.
Used for making pharmaceuticals; as
a warning gas in poisonous, odorless gasses; as a tear gas
If this chemical gets into the eyes, remove anycontact lenses at once and irrigate immediately for at least15 min, occasionally lifting upper and lower lids. Seek medical attention immediately. If this chemical contacts theskin, remove contaminated clothing and wash immediatelywith soap and water. Seek medical attention immediately. Ifthis chemical has been inhaled, remove from exposure,begin rescue breathing (using universal precautions, including resuscitation mask) if breathing has stopped and CPR ifheart action has stopped. Transfer promptly to a medicalfacility. When this chemical has been swallowed, get medical attention. Give large quantities of water and inducevomiting. Do not make an unconscious person vomit.Medical observation is recommended for 24- 48 h afterbreathing overexposure, as pulmonary edema may bedelayed. As first aid for pulmonary edema, a doctor orauthorized paramedic may consider administering a corticosteroid spray
Color Code—Blue: Health Hazard/Poison: Storein a secure poison location. Prior to working with thischemical you should be trained on its proper handling andstorage. Store in tightly closed containers in a cool, well-ventilated area away from oxidizers, strong acids, strongbases, reducing agents, heat, and sources of ignition. Wherepossible, automatically pump liquid from drums or otherstorage containers to process containers.
UN1603 Ethyl bromoacetate, Hazard class: 6.1;
Labels: 6.1-Poisonous materials.
Wash the ester with saturated aqueous Na2CO3 (three times), 50% aqueous CaCl2 (three times) and saturated aqueous NaCl (twice). Dry with MgSO4, CaCl2 or CaCO3, and distil it. [Beilstein 2 IV 527.] LACHRYMATORY.
Vapor may form explosive mixture with
air. Incompatible with oxidizers (chlorates, nitrates, peroxides, permanganates, perchlorates, chlorine, bromine, fluorine, etc.); contact may cause fires or explosions. Keep
away from alkaline materials, strong bases, strong acids,
oxoacids, epoxides, and reducing agents. Esters are generally incompatible with nitrates. Moisture may cause hydrolysis or other forms of decomposition.