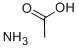Ammonium acetate is a white, deliquescent crystalline solid with a slightly acetic odor. It is used as a reagent in analytical chemistry for determining lead and iron content,a chemical intermediate in manufacturing acetamide. It is also a diuretic in veterinary medicine, in other drugs, in textile dyeing, in meat preservative, in foam rubbers, in vinyl plastics, in stripping explosives, and in determining lead and iron content.

White crystalline solid; deliquescent; melts at 114°C; decomposes at elevated temperatures; density 1.17 g/cm3 at 20°C, density of a 10% solution 1.022 g/mL, and a 50% solution 1.092 g/mL; very soluble in cold water (1,480 g/L at 4°C); also soluble in cold alcohol and acetone (78.9 g/L in methanol at 15°C); solution loses ammonia on standing and becomes acidic.
Ammonium acetate solution can be used to study molecular biology, biological buffers, reagents and DNA and RNA purification. Ammonium acetate solution has been used to study pharmacokinetic analysis of α and β epimers of glycyrrhetinic acid in rat plasma. Ammonium acetate solution has also been used in a study to develop a method for the simultaneous determination of aristolochic acids A and B in some Chinese herbals and traditional Chinese patent medicines.
Ammonium acetate is a reagent used in chromatographic analysis of various compounds such as oligos, proteins, and peptides. It is also employed with acetic acid to provide a buffer solution. Generally it behaves as a catalyst in many reactions like Knoevenagel, Borch. It is also employed as a nutrient additive.
Ammonium acetate is used in the manufacture of acetamide and as a diuretic and diaphoretic in medical applications. The wool industry also uses this salt as a dye mordant.
Buffer solution; determination of lead and iron; separating lead sulfate from other sulfates.
ChEBI: Ammonium acetate is an ammonium salt obtained by reaction of ammonia with acetic acid. A deliquescent white crystalline solid, it has a relatively low melting point (114℃) for a salt. Used as a food acidity regulator, although no longer approved for this purpose in the EU. It has a role as a food acidity regulator and a buffer. It is an acetate salt and an ammonium salt.
Ammonium acetate is manufactured by neutralizing acetic acid with ammonium carbonate or by passing ammonia gas into glacial acetic acid. Acidic ammonium acetate, CH3CO2NH4.CH3CO2H[25007-86-7], is manufactured by dissolving the neutral salt in acetic acid.
A white crystalline solid. The primary hazard is the threat to the environment. Immediate steps should be taken to limit its spread to the environment. Ammonium acetate is used in chemical analysis, in pharmaceuticals, in preserving foods, and for other uses.
Ammonium acetate causes the decomposition of sodium hypochlorite within a few seconds [Mellor 2 Supp. 1:550 1956].
Inhalation of dust irritates nose and mouth. Ingestion irritates mouth and stomach. Contact with dust causes irritation of eyes and mild irritation of skin.
Special Hazards of Combustion Products: Irritating vapors of ammonia acetic acid, and nitrogen oxides may form in fires.
Flammability and Explosibility
Non flammable
Ammonium acetate can be used in proteinaceous baits and in beer waste to increase their attraction to Ceratitis capitate. It helps in the progression of acetone-butanol-ethanol (ABE) fermentation when it is used as the only nitrogen source.
Poison by intravenous route. Moderately toxic by intraperitoneal route. When heated to decomposition it emits toxic fumes of NO, and NH3
Ammonium acetate is used as a chemical
reagent, to make drugs; foam rubber; vinyl plastics;
explosives, and to preserve foods. An environmental threat.
UN3077 Environmentally hazardous substances,
solid, n.o.s., Hazard class: 9; Labels: 9-Miscellaneous hazardous
material, Technical Name Required.
Crystallise it twice from anhydrous acetic acid, and dry under vacuum for 24hours at 100o [Proll & Sutcliff Trans Faraday Soc 57 1078 1961].
Combustible solid. Incompatible with
oxidizers (chlorates, nitrates, peroxides, permanganates,
perchlorates, chlorine, bromine, fluorine, etc.); contact may
cause fires or explosions. Keep away from alkaline materials,
strong bases, strong acids, oxoacids, epoxides, sodium
hypochlorite, potassium chlorate, sodium nitrite.