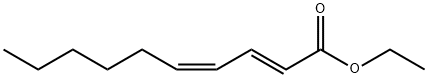
Ethyl (2E,4Z)-2,4-Decadienoate has been identified in pears and has the
typical aroma of Williams pears. Synthesis of ethyl (2E,4Z)-2,4-decadienoate
starts from (Z)-1-heptenyl bromide, which is converted into a 1-heptenyllithium
cuprate complex with lithium and copper iodide. Reaction with ethyl propiolate
yields a mixture of 95% ethyl (2E,4Z)- and 5% ethyl (2E,4E)-2,4-decadienoate.
Pure ethyl (2E,4Z)-2,4-decadienoate is obtained by fractional distillation. A
biotechnological process for its preparation has been developed.
clear colorless to yellowish liquid
Ethyl-trans-2, cis-4-decadienoate has a characteristic pear-like flavor and a light fruity aroma.
Reported found in Bartlett pears, apple, grape, durian (Durio zibethinus), pear brandy, quince and spineless
monkey orange (Strychnos madagasc).
Potential applications include the profile enhancement of guava, melon, pear, apple, banana, kiwi, grape, golden delicious apple, fruit cocktail juices, fruit nectar, mamey and other tropical backgrounds.
ChEBI: Ethyl (2E,4Z)-deca-2,4-dienoate is a fatty acid ethyl ester resulting from the formal condensation of the carboxy group of (2E,4E)-deca-2,4-dienoic acid with the hydroxy group of ethanol. It has a role as a plant metabolite, a flavouring agent, a fragrance and a kairomone. It is functionally related to a (2E,4E)-deca-2,4-dienoic acid.
Detection: 100 ppb. Aroma characteristics at 1.0%: sweet ripe pear, creamy and slightly fatty with fruity,
green, waxy apple, fleshy nuances
Taste characteristics at 5 ppm: ripe pear, green fruity, waxy apple, tropical notes, fatty golden delicious apple
fleshy. Taste characteristics at 20 ppm: green, fruity, apple and pear with waxy tropical nuances.
Natural occurrence: Apple, pear, grape, pear brandy and quince.
Synthetically via the lithium vinyl cuprates.