Off-white to salmon crystalline powder
Bromoxynil is a colorless to white crystalline
solid or tan powder. Odorless (pure).
Selective contact foliage-applied herbicide used to control many broad-leaved weeds
in cereals.
Bromoxynil is a nitrile herbicide. Bromoxynil is effective in post-emergent control of broad leaf weeds in cereal, corn, sorghum, onions, flax, mint, turf, and on non-cropland.
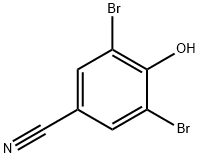
ChEBI: A dibromobenzene that is 2,6-dibromophenol substituted by a cyano group at position 4.
Colorless solid. Melting point 382-384°F (194-195°C). Sublimes at 275°F (135°C) under pressure of 0.15 mm Hg. Used as a herbicide.
Bromoxynil is a weak acid.
Herbicide: A U.S. EPA restricted Use Pesticide (RUP). For
post-emergent control of broadleaf weeds. Used on alfalfa,
garlic, corn, sorghum, flax, cereals, turf and on pasture and
rangelands.
BRIOTRIL®; BRUCIL®; BRITTOX®;
BROMINAL®; BROMINEX®; BROMINAL®;
BROMINAL ME-4®; BROMINIL®; BROMILIL
PLUS®; Bromox 2E; BROMOTRIL®; BROMOXYNIL
NITRILE HERBICIDE®; BRONATE®; BROXYNIL®;
BUCTRIL® Bromoxynil; BUCTRIL® GEL HERBICIDE
(octanoate); BUCTRIL® 4EC GEL (mixture of bromoxynil
octanoate + bromoxynil heptanoate); BUCTRIL
INDUSTRIAL®; CHIPCO BUCTRIL®; CHIPCO
CRAB-KLEEN®; FLAGON®, 400 EC; HOBANE®;
LABUCTRIL®; LITAROL®; M&B 10064®; MB
10064®; MB 10731® (octanoate); M&B 10731®; ME4
BROMINAL®; MERIT®; MEXTROL-BIOX®; MOXY
2E®; NCR CE EE DOV7® (octanoate); NU-LAWN
WEEDER®; OXYTRIL M®; PARDNER®; SABRE®;
TORCH®
Bromoxynil is a hydroxybenzonitrile
herbicide used for postemergent control of broadleaf
weeds; on alfalfa, garlic, corn, sorghum, flax, cereals,
turf and on pasture and rangelands. A United States
Environmental Protection Agency RUP.
Biological. Duke et al. (1991) reported that bromoxynil can be converted to 3,5-
dibromo-4-hydroxybenzoic acid by a microbial nitrolase.
Soil. In soils, Klebsiella pneumoniae metabolized bromoxynil to 3,5-dibromo-4-
hydroxybenzoic acid and ammonia (McBride et al., 1986). In soil, bromoxynil undergoes
nitrile and then amide hydrolysis yielding 3,5-dibromo-4-hydroxybenzoic acid and
Nitrification in soils is inhibited when bromacil is applied at a concentration of <50
ppm (Debona and Audus, 1970). The half-life in soil is approximately 10 days (Hartley
and Kidd, 1987).
Plant. In plants, the cyano group is hydrolyzed to an amido group which is subsequently
oxidized to a carboxylic acid. Hydrolyzes to hydroxybenzoic acid (Hartley and Kidd,
1987). In plants, bromoxynil may hydrolyze to a benzoic acid (Humburg et al., 1989).
Bromoxynil-resistant cotton was recently developed by inserting a bxn gene cloned from
the soil bacterium Klebsiella ozaenae. This gene, which encodes a specific nitrolase,
converted bromoxynil to its primary metabolite 3,5-dibromo-4-hydroxybenzoic acid
(Stalker et al., 1988).
Chemical/Physical. Emits toxic fumes of nitrogen oxides and bromine when heated
to decomposition (Sax and Lewis, 1987). Reacts with bases forming water-soluble salts
(Worthing and Hance, 1991). Bromacil is stable to UV light (Hartley and Kidd, 19
UN2588 Pesticides, solid, toxic, Hazard Class:
6.1; Labels: 6.1—Poisonous materials, Technical Name
Required.
A weak acid; keep away from bases and
alkalies. React with boranes, alkalies, aliphatic amines,
amides, nitric acid, sulfuric acid. Keep away from oxidizers
(chlorates, nitrates, peroxides, permanganates, perchlorates,
chlorine, bromine, fluorine, etc.) and strong acids.
Do not discharge into drains
or sewers. Dispose of waste material as hazardous waste
using a licensed disposal contractor to an approved landfill.
Incineration with effluent gas scrubbing is recommended.
Consult with environmental regulatory agencies for guidance
on acceptable disposal practices. If allowed,
Incineration with effluent gas scrubbing is recommended.
Containers must be disposed of properly by following
package label directions or by contacting your local or federal
environmental control agency, or by contacting your
regional EPA office.