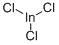Indium chloride is a useful catalyst for aqueous organic reactions including C-C bond formation, aldol reaction, Friedel-Crafts acylation, Diels-Alder reaction and reduction1-4. It is an efficient, mild, Lewis acid for organic synthesis. It is an efficient catalyst for thioacetalization and its chemoselectivity. It can also catalyze the enantioselective ring-opening of meso-aziridines with thiols. Moreover, it can act as a highly efficient catalyst in water for the preparation of furyl glycosides.
Catalyzes the enantioselective ring-opening of meso-aziridines with thiols.1,2 A highly efficient catalyst in water for the preparation of furyl glycosides.3
Indium(III) chloride is used as catalyst for aqueous organic reactions including C-C bond formation, aldol reactions, and reductions and for organic synthesis.
In electroplating using a solution of the salt with dextrose and NaCN. This solution is stable, though it turns dark on standing and deposits a mud which, however, contains no indium.
Indium(III) chloride (InCl3) is an indium halide that is a Lewis acid in a variety of organic reactions.
The anhydrous salt forms yellow deliquescent crystals which can be sublimed at 600o in the presence of Cl2/N2 (1:1) [does not melt]. It is resublimed in the presence of Cl2/N2 (1:10) and finally heated to 150o to expel excess Cl2. It is soluble in H2O and should be stored in a tightly closed container. [Baxter & Alter J Am Chem Soc 55 1943 1933.]